Pfizer and BioNTech have developed a COVID-19 vaccine they claim has 95 per cent efficacy. But Australia's medical research bodies haven't given up, despite the difficult funding and regulatory environment.
At the height of summer, as Australians remained preoccupied by bushfires, a visitor to Melbourne from Wuhan in China was rushed to hospital with a fever, a cough and breathing difficulties.
At 2am on 25 January, virologists from the Peter Doherty Institute for Infection and Immunity — a collaboration between the University of Melbourne and the Royal Melbourne Hospital — confirmed Australia’s first case of what the World Health Organization (WHO) would come to call COVID-19.
Little more than 48 hours later, having introduced a sample of the patient’s sputum into a culture of monkey kidney cells, those scientists became the first to grow the new coronavirus in a laboratory outside of China — an important step towards improving the accuracy of tests, developing vaccines and screening existing antiviral drugs.
It’s important to recall that most Australians remained oblivious to the looming threat at that stage. But researchers around the country had been predicting and preparing for a pandemic for years; they knew the best chance of beating it was with all hands on deck. So, despite the competitive nature of contemporary research environments, the Doherty Institute shared phials of the virus with WHO and public health laboratories across the globe. This act reflected not only the high quality of the medical research community in Australia, but also the foresight and collaborative efforts of a range of institutions in responding to the health crisis.
Collaboration, both internally and externally, has never been more crucial than in the current climate, says Sharon Lewin AO, director of the Doherty Institute and a professor of medicine at the University of Melbourne. “This is not business as usual. Sharing your knowledge is hugely important in a pandemic response,” she says.
Lewin points to several projects in which Doherty Institute researchers are working with industry partners and their counterparts at other medical research institutes to evaluate antiviral drugs, explore the potential for antibody-related therapies and uncover the impacts of quarantine and isolation.
For example, a consortium of researchers at the Doherty Institute, Walter and Eliza Hall Institute of Medical Research, CSL, Affinity Biosciences, Commonwealth Scientific and Industrial Research Organisation (CSIRO), Burnet Institute and Kirby Institute are exploring antibodies that block the “lock and key” mechanism through which the coronavirus binds to human lung cells — disrupting the first step in the infection cycle.
Traditional barriers between scientific and professional staff members are also dissolving, with COVID-19 forcing new ways of working, says Professor Doug Hilton AO, director of Melbourne’s Walter and Eliza Hall Institute. “The pandemic has accelerated the cohesiveness of our teams… and that’s been a positive part of our response,” he says.
Longer-term viability
Early in the pandemic, as medical research institutes grappled with immediate challenges, such as establishing strict safety protocols for laboratory work and transitioning staff to work from home, they also kept one eye on what lay ahead.
In February, the Florey Institute of Neuroscience and Mental Health established a COVID-19 taskforce comprising a cross-section of leaders who liaised with the institute’s board members and sought their understanding of how risk might affect the institute. “The board helped keep the taskforce team focused on the long-term view,” says Professor Steven Petrou, director of the Florey.
“As one example, very early on when the risk that COVID-19 presented was still uncertain, the board encouraged development of a business continuity plan with multiple scenarios about what the future might look like. This has been a valuable document and provided a strong framework for us to... respond rapidly to change in our external environment.”
According to Professor Brendan Crabb AC, a director and CEO at the Burnet Institute, a similar COVID-19 initiative was formed “to make sure that the day-to-day emergency isn’t the only thing we’re responding to”.
Like its counterparts, the Burnet Institute largely shifted its focus to COVID-19-specific projects targeting the global response, which means that other research has, by necessity, been put on hold. About half of the institute’s staff and students are now engaged in work related to COVID-19, compared to the Doherty Institute’s 30 per cent.
Crabb says funders, recognising the enormity of the emergency, have typically allowed flexibility in the use of grants that were intended to be used for something other than COVID-19.
Burnet Institute chair Mary Padbury GAICD says that some of the board’s focus has inevitably turned to the financial and other impacts of this shift. “Recognising that some sources of funding have asked us to do particular bits of work, which we won’t be able to, where does that leave the institute’s financials?” she says. “We have been thinking about those slightly longer-term challenges to ensure we’re not taking our eye off some of those important sustainability balls.”
According to Crabb, other pressing health issues will also be impacted. For example, Australia’s COVID-19 travel restrictions have affected the Burnet Institute’s malaria-related work in Papua New Guinea. “Our concern is that the non-COVID-19 health consequences will far outweigh the COVID-19-specific health consequences — within one malaria season, you can undo 10 years of good work,” he says.

Governance challenges
At the Brisbane-based University of Queensland (UQ), the determination to capitalise on past discoveries united disparate parts of the organisation. These include the governing 22-member senate, technology transfer company UniQuest (responsible for commercialising UQ research) and multiple departments — from fundraising and finance to communications and legal — says Dr Sally Pitkin FAICD, a UQ senator who served for a decade on the board of UniQuest.
“UQ researchers developed this molecular clamp back in 2011, but no-one was interested in it at the time, because no-one could see any commercial benefit,” she explains. “But, as it turns out, a molecular clamp is particularly relevant for pandemic vaccines.”
Pitkin said UQ could have supplied its molecular clamp technology to the Coalition for Epidemic Preparedness Innovations (CEPI) — a low-risk strategy that would still have garnered reputational benefits for the university. Instead, the seriousness of the health crisis was such that UQ chose to forge ahead and develop a vaccine based on the foundation it had already built, despite considerable additional risks and costs. (UQ’s coronavirus vaccine is considered by experts to be a leading candidate.)
“Significant medical research involves extraordinarily complex contractual arrangements and very sophisticated frameworks to manage risk,” says Pitkin. “In relation to the development of this vaccine, there are 95 different contractual arrangements in place — all of which needed to be negotiated and agreed — so you can imagine how many parts of the university were involved.
“But it’s a time where universities and medical research institutes have to step up and be counted,” she adds.
Ensuring correct processes were followed when things were changing so fast and affecting so many presented unique governance challenges. Typically, vaccine development programs run sequentially, so researchers wait for the results of Phase 1 trials before committing any funds to scaling up the process. But with COVID-19’s grim toll growing by the day, “there wasn’t time to do that”, says Pitkin. Risk appetites increased because of what’s at stake.
So as scientists got on with the job, other parts of the tertiary institution swung into action, raising $23m to ensure that if Phase 1 trials were successful, then the next phase could proceed without delay.
It was also important that scientists be “quarantined from the sense of urgency” driving other elements of decision-making, she says. “There can be no shortcuts in medical research — the ethics, safety and efficacy of what you’re trying to develop has to remain paramount.”
Future funding crunch
The COVID-19 crisis galvanised the rapid release of new funding by state and federal governments and philanthropic agencies. In June, for instance, the Morrison government announced $66m towards the search for a vaccine and treatments, in addition to the $30m already pledged.
Lewin estimates that in the current environment, about 20 times more funding is available for research directed towards COVID 19 compared to non-COVID research.
The nation’s Medical Research Future Fund has, over the past five or more years, had a massive impact on funding of translational health and medical research.
However, as the pandemic continues to cause economies to contract, businesses to fail and discretionary income to dwindle, the ongoing commitment to the growth of research funding by governments remains to be seen.
Professor Scott Bell, CEO of the Queensland-based Translational Research Institute (TRI), says there is considerable uncertainty about university and healthcare funding into the future.
Former TRI chief executive (2010–14), chair of the TRI Foundation board and one of the higher-profile researchers associated with the institute is Professor Ian Frazer FAICD, who — along with the late molecular biologist and virologist Dr Jian Zhou — developed Gardasil, the vaccine for the cervical-cancer-causing human papillomavirus (HPV).
Bell described the development of this vaccine as a stand-out example of how the commercialisation of research findings can ultimately lead to better healthcare outcomes for people around the world. “The health and medical research community in Australia have an exceptional track record in scientific excellence,” he says.
But a decline in Australia’s manufacturing capabilities since the 1970s, and at the time the HPV vaccine was approaching large-scale clinical trials, meant it was unable to be tested and manufactured locally. Instead, the trials were conducted overseas with the support of large pharmaceutical companies such as the US-based Merck.
“Rather than selling our discoveries, we should be growing our advanced health and medical manufacturing capability in order to leverage off our excellence in the university and medical research institutes across Australia,” says Bell.
He points out that it’s commonplace in parts of Europe and North America to see manufacturing companies embedded in notable universities.
This is “just part of the process” at places such as Germany’s University of Tübingen and Toronto’s MaRS Discovery District innovation hub, where the University of Toronto was a founding partner.
“As researchers,” says Bell, “we need to think more laterally towards addressing questions such as what are the commercial opportunities, what contract research can be undertaken and what are the opportunities to grow what we are working on in our research programs to support the development of translational products?”
Bell explains that TRI accommodates half-a-dozen startups that “could fold at any moment, given their dependence on grant funding and venture capital”. But when these companies make valuable discoveries, they rapidly outgrow Australia’s capacity to meet their needs in terms of undertaking good manufacturing practice and the early phases of trials. Vaxxas, a startup-in-residence at TRI since 2014, shortly after TRI was founded, is developing a “game-changing” platform for the delivery of vaccinations. It applies a needle-free NanopatchTM to the skin to deposit the vaccine among immune cells. This technology has the added benefit of not requiring refrigeration.
“This is one of the companies that we would like to continue to support,” says Bell. “We don’t want to sell off the intellectual property too quickly just because we don’t have the capability to support the further clinical trial development and manufacturing of such potential products.
When [they are] proved effective in five years’ time, we’re going to have to buy those treatments, such as vaccinations, back — just as we did with Gardasil.”
TRI has engaged in discussions with state and federal governments to explore funding opportunities for TRI2, a purpose-built scale-up and advanced manufacturing facility for the next stage of development of startup medical technology companies.
“Over the past two years, the board has been key in supporting the steering group for the TRI2 detailed business case submission to governments in early September [2020],” says Bell. “Specifically, they have supported the development and financing of the detailed business case for TRI2 and also worked with their connections at both levels of government.”
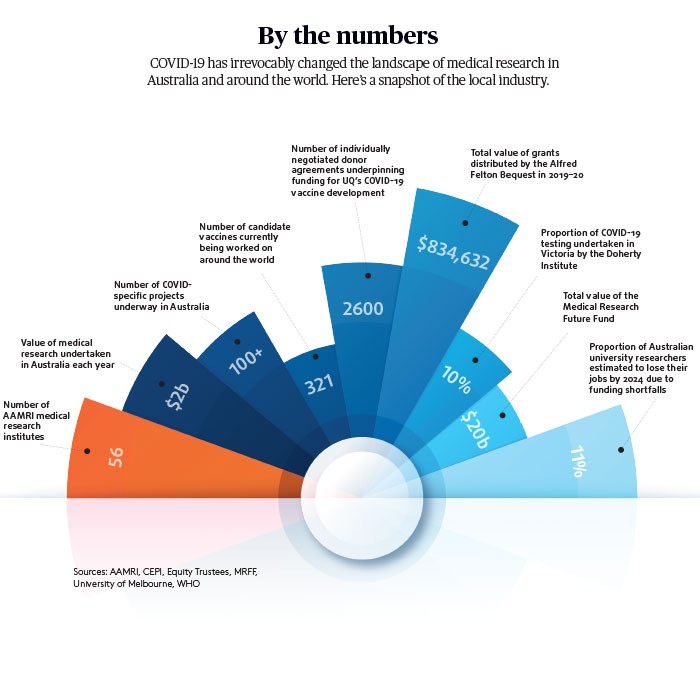
Across the research sector, alarm bells are ringing in relation to the foreshadowed loss of between 5100 and 6100 university researchers (around 11 per cent) by 2024. A paper by the University of Melbourne attributes this to the sharp drop in discretionary income caused by the lack of international full-fee paying students.
Already, many promising postdoctoral researchers face barriers in funding the next stage of their career development. “It’s often seen as a bottleneck, with many having to consider changing career paths to work predominantly in an educator or teaching role, or leaving our shores to pursue research careers where research funding is more readily available,” says Bell. “Being able to diversify opportunities for these talented researchers through career paths into industry is clearly important.”
Investing in the future of medical research
The federal government’s Medical Research Future Fund (MRFF) seeks to underpin the development of life-changing health and medical discoveries including, more recently, responses to the COVID-19 crisis.
Established in 2015 and predicted to grow to more than $21 billion by 2022–23, the MRFF invests in Australian research at all stages, from idea generation through to laboratory trials and onto the development of the final commercial product.
According to the Association of Australian Medical Research Institutes (AAMRI), the Morrison government bolstered the fund by $3.2b at the start of the financial year — almost $700m more than forecast in the previous year’s budget. This will drive nearly $580m in investment in medical research in 2020–21, growing to $645m in 2021–22 and $650m each year thereafter, notes AAMRI.
Funds, allocated according to national research priorities, are designed to help researchers develop their ideas locally and boost the country’s burgeoning biotechnology industry.
To date, the MRFF has allocated a total of $95.2m to COVID-19 research. This includes $19m for vaccine development, $12.7m for antivirals, $6.8m for respiratory health medicine, $3.6m for diagnostics and $1.8m for public health.
The 2020–21 Budget injected a further $1b in research funding for universities, provided through the federal government’s Research Support Program. This one-off measure is designed to help universities plug the financial hole caused by the marked decline in international higher education revenue.
Several smaller spending announcements in the budget include funds for preventative health initiatives, vaccine rollout in the region and mental health research.
Funds were also set aside for research infrastructure through the National Collaborative Research Infrastructure Strategy, including investment in a new synthetic biology research infrastructure facility.
From 2020–21, an extra $459.2m over four years will also be provided to the CSIRO.
Latest news
Already a member?
Login to view this content